Cutting-edge Sharing | Therapeutic Potential And Industrialization Process Of Exosomes
Dec 12, 2025
Leave a message
The Intelligent Messengers of Cells-Exosomes
All cells, both prokaryotes and eukaryotes, secrete extracellular vesicles (EVs). Among them, exosomes are EVs with a diameter of 30–150 nm, featuring a bilayer membrane structure. They carry DNA, RNA, proteins, lipids, and metabolites, functioning in intercellular communication and signal transduction.
(Note: EVs include microvesicles and exosomes. However, due to the high structural and compositional similarity between microvesicles and exosomes, as well as the inherent heterogeneity of EVs themselves, it is nearly impossible to strictly distinguish whether a specific EV is a microvesicle or an exosome in practice. Currently, in China's industrial and scientific research communities, "exosomes" is the most commonly used term when referring to specific therapeutic applications.)
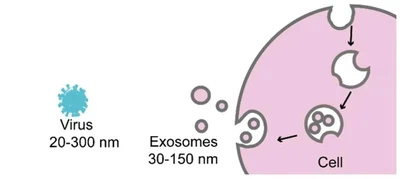
Source: Exosome Theranostics: Biology and Translational Medicine (Theranostics)As illustrated, exosomes are extracellular vesicles originating from endocytosis, with a size comparable to that of virions.
Exosomes have garnered high expectations primarily due to their diverse functions. Exosomes from different sources vary in their cargo, and they are generally recognized to reflect the physiological status and functional characteristics of their parent cells-a feature that endows exosomes with the ability to play diverse roles in intercellular communication. For instance, via the specific miRNAs and proteins they carry, exosomes play crucial roles in various contexts such as cardiovascular protection, bone repair, immune regulation, and skin regeneration.

Source: Exosome-derived noncoding RNAs in gastric cancer: functions and clinical applications. Mol CancerShown are the signature characteristics and internal components of exosomes.
The key turning point in industrial development
Despite broad prospects, the exosome field still faces challenges in the early stage of industrialization, with global quality standards and safety evaluation systems yet to be improved. Against this backdrop, China has significantly accelerated its regulatory progress - in June this year, the Center for Drug Evaluation (CDE) of the National Medical Products Administration (NMPA) issued the Scope, Classification and Interpretation of Human Gene Therapy Products, Cell Therapy Products and Advanced Therapy Medicinal Products (Draft for Comment), formally incorporating extracellular vesicles (EVs) into the regulatory scope of "Advanced Therapy Medicinal Products (ATMPs)" for the first time; subsequently in September, the State Council officially promulgated the Regulations on the Administration of Clinical Research and Clinical Translation Application of New Biomedical Technologies, further establishing a clear dual-track transformation path of "drug" and "technology" for innovative products such as exosomes. The successive introduction of these policies not only marks that exosome therapy has officially entered a new stage of standardized and high-quality development but also provides a solid institutional guarantee for the industrial transformation from laboratory to clinical application.
Currently, three directions are most promising:
As therapeutic agents: Preliminary clinical studies have demonstrated their ability to promote tissue repair and regeneration in fields such as diabetic foot ulcers, burns, and osteoarthritis.
As drug delivery carriers: Through engineering modification, exosomes can be loaded with anticancer drugs, nucleic acid drugs, etc., and target diseased cells, holding broad prospects in cancer therapy, neurological diseases, and other fields.
As disease diagnostic tools: Exosomes are widely present in body fluids such as blood and urine. By analyzing their cargo, they are expected to enable early and non-invasive diagnosis of diseases such as cancer.
Breakthrough in the large-scale production of exosomes
Whether for research or future therapeutic applications, the prerequisite is obtaining high-quality, high-yield, and batch-homogeneous exosomes. Among these requirements, cell culture plays a crucial role. Traditional cell culture methods utilize fetal bovine serum (FBS), but the exogenous exosomes and complex components inherent in FBS severely interfere with the purity and safety of the final product. Additionally, there is a risk of animal-derived pathogens, making it unable to meet clinical-grade production requirements. The ideal solution is to use specialized serum-free media that are chemically defined and xeno-free.
The Exo MSC CD Exosome Expression Kit is a serum-free, xeno-free, and chemically defined culture system specifically designed by BioEngine for MSC culture during exosome production. It aims to provide a stable and efficient cell culture solution for exosome drug development and large-scale manufacturing.

(With Phenol Red) / (Without Phenol Red) Exo MSC CD Exosome Expression Kit
Product Advantages:
Significantly enhances yield and quality: Exosome yield is increased by 2.8-fold while maintaining stable MSC expansion; exosome quality is improved to ensure functional integrity.
Improves process stability and purification efficiency: Serum-free and chemically defined formulation eliminates batch-to-batch variation, enhancing process stability; greatly simplifies the purification process and improves recovery efficiency.
End-to-end serum-free solution: Compatible with the Omni-Expansion MSC Culture Kit, enabling end-to-end serum-free culture from MSC isolation and passage to exosome production with high compliance.
Product Performance:
1.Exosome yield increased by 2.1-fold and 2.8-fold respectively, significantly reducing production costs:After seeding MSCs and allowing them to adhere (for at least 24 hours), replace the medium with the Exo MSC CD Exosome Expression Kit. Subsequently, harvest the supernatant every 48 hours and replenish with fresh medium to achieve continuous and efficient exosome collection.In the Exo MSC CD Exosome Expression Kit, the exosome yield reaches 1.6×10¹⁰ particles/mL, which is 2.1-fold and 2.8-fold higher than that of Brand A (7.8×10⁹ particles/mL) and Brand B (5.7×10⁹ particles/mL), respectively. Additionally, particle size analysis shows that the main peak of the particle size distribution is at 121.6 nm, accounting for 97.1% of the total-consistent with the typical exosome particle size range (30–150 nm).


2.High-quality exosomes with typical morphological and phenotypic characteristics: Transmission Electron Microscopy (TEM) results show that the extracted vesicles exhibit a clear cup-shaped biconcave disc structure, presenting the typical morphology of exosomes.
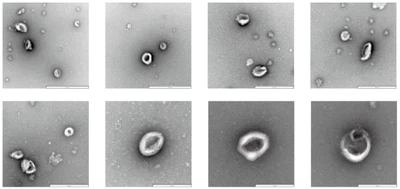
Electron micrograph of exosomes
Western Blot results showed that exosomes collected at the first (1D2) and second (2D2) time points both exhibited the typical expression profile of exosomal protein markers: TSG101, CD63, and CD9 were positively expressed, while Calnexin was negatively detected.

Throughout the entire culture process, MSCs maintained a typical fibroblast-like morphology, indicating a stable cell state.


