The Evolution Of Influenza Vaccine Production: MDCK Cell-Driven, Breaking The Constraints Of Chicken Embryos
Jan 29, 2026
Leave a message
Manufacturing Process of Influenza Virus Vaccines
Recently, respiratory diseases have entered a high-incidence period, with a superposition of multiple pathogens including influenza, COVID-19, mycoplasma, RSV and adenoviruses, among which influenza is the predominant one. According to the data from the Weekly Influenza Surveillance Report (Week 47, 2023: November 20–26, 2023) compiled by China National Influenza Center, the proportions of influenza-like illness (ILI) cases in southern and northern provinces reached 8.0% and 7.1% respectively, which were significantly higher than the figures for the same period in previous years (see the table below) and showed an upward trend (see the figure below).
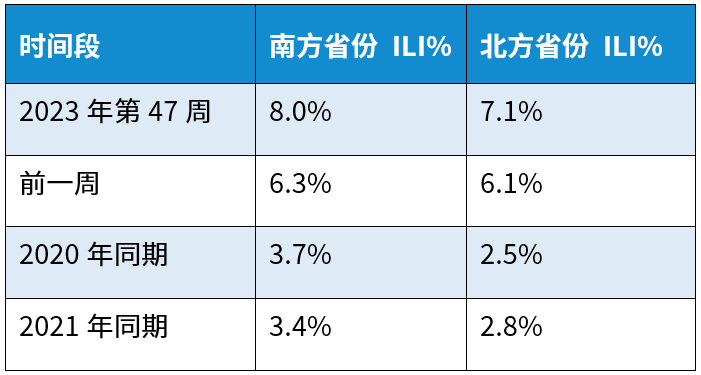

Source: Weekly Influenza Surveillance Report, Issue 47
Meanwhile, laboratory tests on influenza-like illness (ILI) samples have shown a continuous rise in the positive detection rate of influenza viruses, with the rates standing at 44.3% in southern provinces and 36.9% in northern provinces respectively. A(H3N2) subtype has been the predominant strain in both regions, followed by the B(Victoria) lineage (see the table and figure below).
Laboratory Testing for Influenza-like Illness (ILI) Surveillance
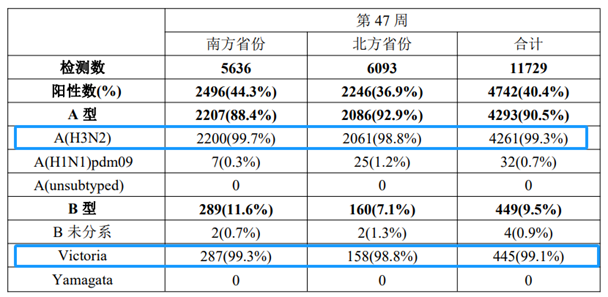
Source: Weekly Influenza Surveillance Report, Issue 47
Test Results of Influenza-like Illness (ILI) Specimens in Southern Provinces

Influenza Vaccines
Influenza is an acute respiratory infectious disease caused by influenza viruses, to which the general population is universally susceptible. Annual seasonal influenza epidemics result in 3 million to 5 million severe cases and 290,000 to 650,000 respiratory disease-related deaths worldwide, posing a severe threat to human health. Based on differences in nucleoproteins and matrix proteins, influenza viruses are mainly classified into types A, B, C and D, all of which can infect humans. Influenza A viruses are further divided into multiple subtypes according to the protein structures and genetic characteristics of hemagglutinin (HA) and neuraminidase (NA) on the viral surface. To date, 18 HA subtypes (H1-H18) and 11 NA subtypes (N1-N11) have been identified. Influenza B viruses are categorized into the Victoria lineage and Yamagata lineage based on HA genotypes. Since March 2020, naturally circulating influenza B/Yamagata lineage strains have been barely detected globally. As a result, the influenza viruses currently causing epidemics are mainly the H1N1 and H3N2 subtypes of influenza A virus, and the Victoria lineage of influenza B virus.
Vaccination is the most cost-effective measure to prevent influenza and reduce the burden of influenza-related severe illness and death. Influenza vaccines currently marketed worldwide include inactivated vaccines, live attenuated vaccines and recombinant vaccines. Classified by their components, they are divided into trivalent and quadrivalent influenza vaccines. Trivalent influenza vaccines contain one lineage of influenza A(H3N2) subtype, A(H1N1)pdm09 subtype and influenza B virus, while quadrivalent influenza vaccines contain influenza A(H3N2) subtype, A(H1N1)pdm09 subtype, as well as influenza B(Victoria) lineage and B(Yamagata) lineage. The influenza vaccines used in China for the 2023-2024 epidemic season include trivalent inactivated influenza vaccine (IIV3), quadrivalent inactivated influenza vaccine (IIV4) and trivalent live attenuated influenza vaccine (LAIV3).
Manufacturing Processes of Influenza Vaccines
Limitations of Traditional Egg-based Influenza Vaccine Production
Influenza vaccine production processes include egg-based and cell-based culture methods. At present, the most widely used manufacturing process for influenza vaccines is egg-based influenza virus culture, which has limitations in terms of temporal flexibility and vaccine yield. Egg-based production is associated with drawbacks such as unstable raw material supply and long production cycles. It is difficult to produce billions of vaccine doses in a short period using egg-based methods, and shortages will also occur for other vaccines manufactured via this approach.
Cell-based Influenza Vaccine Production: An Emerging Trend
Compared with traditional egg-based processes, cell culture production systems can manufacture large quantities of vaccines in a short time and significantly enhance human capacity to respond to pandemics and emerging viral diseases. For example, the preparation of a single dose of egg-based vaccine requires 1-2 fertilized chicken eggs. That is to say, large-scale influenza vaccine production demands at least millions of fertilized embryonated eggs, all of which need to be prepared in advance. In contrast, in cell-based production processes, cells as starting materials can be preserved and used on demand at any time.
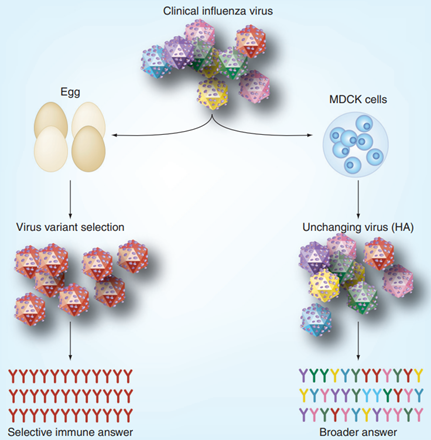
Safety of MDCK cell culture-based influenza vaccines,
Future Medicine, 2011
In addition, influenza viruses cultured in eggs may undergo egg-adaptive gene mutations, including mutations in the antigenic hemagglutinin (HA). While such mutations facilitate viral infection and replication in eggs, they result in discrepancies between the egg-cultured virus and the original clinically isolated strain, which may compromise vaccine efficacy. In contrast, no cell-specific adaptive mutations have been observed in Madin–Darby canine kidney (MDCK) cells, and the viruses cultured in MDCK cells are consistent with the original clinically isolated strains, thus ensuring vaccine efficacy (see the figure above). For these reasons, animal cell culture for influenza vaccine production has garnered increasing attention and application, and is gradually replacing the traditional egg-based manufacturing process.
MDCK Cells: One of the Optimal Choices for Influenza Vaccine Production
When selecting a cell line for inactivated influenza vaccine production, the cell line must be susceptible to a broad range of influenza viruses and support high-titer viral replication. MDCK cells are susceptible to all currently known influenza vaccine strains, including avian, swine and equine influenza strains, and enable high viral yield. For this reason, MDCK cells are the preferred choice for most manufacturers of cell-based influenza vaccines. For instance, Solvay, Seqirus (formerly Novartis) and SK Chemicals have successfully launched MDCK cell-based influenza vaccines, which were approved for marketing in the Netherlands (2001), by the US FDA (2016) and in South Korea (2015), respectively. To date, no cell-based influenza vaccines have been approved for marketing in China; however, a number of vaccine manufacturers have laid out the production of MDCK cell-based influenza vaccines, with several candidates having obtained approval for clinical trials (see the figure below).
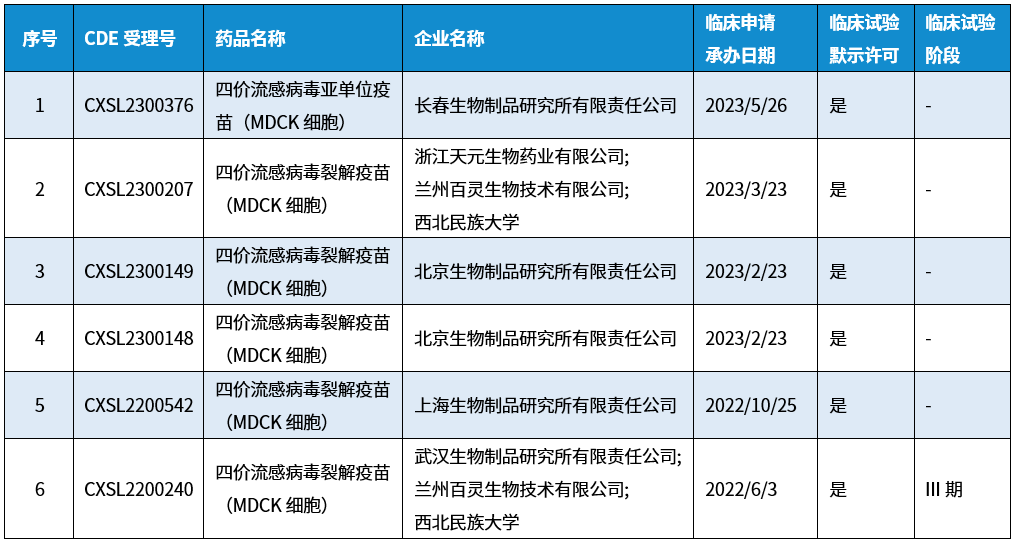
BioEngine's R&D team has been researching MDCK cell culture technology since 1995. Leveraging nearly 40 years of accumulated medium technology expertise and on-site technical service experience at vaccine manufacturing enterprises, we have continuously optimized our MDCK product portfolio and launched the Xeno Series of serum-free MDCK cell culture media. This product line delivers a highly efficient alternative for human influenza vaccine production processes, enabling the transition from traditional egg-based culture to high-efficiency suspension cell culture. We have also achieved a number of innovative breakthroughs both domestically and internationally. For example, as early as 2016, we successfully applied serum-free fully suspended MDCK cell culture technology to avian influenza vaccine production, boosting yields by 6 to 8 times. In recent years, we have supported domestic vaccine manufacturers in adopting this technology for the R&D and production of human influenza vaccines. BioEngine has published numerous papers and obtained patents in the development of serum-free MDCK cell culture media and the design of associated culture processes (see the list of papers at the end of this document for reference).
Published Literature by BioEngine
Focused on the Development of Serum-free MDCK Cell Culture Media and Associated Culture Processes
1. Qian Ye, Thu Phan, Wei-Shou Hu, Xuping Liu, Li Fan, Wen-Song Tan, Liang Zhao. Transcriptomic Characterization Reveals Attributes of High Influenza Virus Productivity in MDCK Cells. Viruses, 2021, 13(11): 22002. Thomas Bissinger, Yixiao Wu, Pavel Marichal-Gallardo.Towards integrated production of an influenza A vaccine candidate with MDCK suspension cells. Biotechnology and Bioengineering, 2021, 118(10): 3996-40133. Yixiao Wu, Xuping Liu, Udo Reichl, Wen-Song Tan. High cell density perfusion process for high yield of influenza A virus production using MDCK suspension cells. Applied Microbiology and Biotechnology, 2021, 105(4): 1-144. Yixiao Wu, Hanjing Jia, Xuping Liu,Wen-Song Tan. Highly Efficient Production of an Influenza H9N2 Vaccine Using MDCK Suspension Cells. Bioresources and Bioprocessing, 2020, 7: 635. Thomas Bissinger, Johannes Fritsch, Adrian Mihut, Yixiao Wu, Xuping Liu, Yvonne Genzel, Wen-Song Tan, Udo Reichl. Semi-perfusion cultures of suspension MDCK cells enable high cell concentrations and efficient influenza A virus production. Vaccine, 2019, 37(47): 7003-70106. Luo Jian, Liu Xu-ping, Xiong Fei-fei, Gao Fei-xia, Yi Ying-lei, Zhang Min, Chen Ze, Tan Wen-song. Enhancing Immune Response and Heterosubtypic Protection Ability of Inactivated H7N9 Vaccine by Using STING Agonist as a Mucosal Adjuvant. Frontiers in Immunology, 2019, 10: 22747. Ding Huang, Wen-Juan Peng, Qian Ye, Xu-Ping Liu, Liang Zhao, Li Fan, Kang Xia-Hou, Han-Jing Jia, Jian Luo, Lin-Ting Zho. Serum-Free Suspension Culture of MDCK Cells for Production of Influenza H1N1 Vaccines. PLOS ONE, 2015, 10(11): e01416868. Ding Huang, Kang Xia-Hou, Xu-Ping Liu, Liang Zhao, Li Fan, Zhaoyang Ye, Wen-Song Tan, Jian Luo, Ze Chen. Rational design of medium supplementation strategy for improved influenza viruses production based on analyzing nutritional requirements of MDCK Cells. Vaccine, 2014, 32(52): 7091-7097

