HIPP Series Serum-Free Media For Immune Cell Culture
Overcome Serum Limitations:
Enable Efficient, Consistent and Scalable Cell Therapy Manufacturing
Traditional serum-containing culture methods are limited by low expansion efficiency, poor viral transduction rates, significant batch-to-batch variability, and elevated contamination risks, making them unsuitable for the production of cell therapy. The HIPP series serum-free media specifically designed for immune cell culture, which enables efficient expansion of immune cells under completely serum-free conditions while maintaining excellent cell viability and functional performanceDescription
Performance
Features

Serum-free, Xeno-free, Chemically defined

Culture process: Static (plates, flasks) & Dynamic (bags, bioreactors)

Specifically designed for primary immune cells (T cells, NK cells, CAR-T, CAR-NK, CIK, TIL, γδT cells, hematopoietic stem cells (CD34+)) and cell lines (NK-92, NK-92MI, K562, Jurkat, etc.)

Performance: High expansion fold, high positive ratio (e.g., CAR+), enhanced memory phenotype, controlled exhaustion phenotype, >95% cell viability
Why Choose HIPP

Superior Growth & Function: High expansion fold & cell viability, desired phenotypes under completely serum-free culture

Seamless Scalability: From small-scale research to large-scale production

Process Flexibility: Cytokine-free for easy protocol customization

Unmatched Consistency & Safety: No serum = Low variability & contamination risk

Regulatory Confidence: GMP-compliant facility (MDSAP (FDA) & ISO 13485:2016 certified) Full regulatory documentation supporting R&D to clinical/commercial production
Proven in Clinical Development

Supported BLA submission for a CAR-T therapy (2024)

Enabled China's first IND-approved neoantigen-targeting T-cell therapy (2019)

Trusted in 20+ cell therapy projects
Order Information
Basal Medium
|
Application |
Product Name |
Cat. No. |
Size |
Form |
Product Instruction (pdf) |
Inquiries /Sample application |
| HIPP-X 100 Kit | HIPP-X 100 Lymphocyte Serum-free Medium (GMP Grade) | EXP0122701 | 1L | Liquid | ||
| HIPP-X 100 Lymphocyte Serum-free Medium Supplement | EXP0122801 | 5mL | Liquid | |||
| HIPP-T009 |
HIPP-T009 Lymphocyte Serum-free Medium (GMP Grade) |
EXP0103801 | 1L | Liquid | ||
| HIPP-T009 Lymphocyte Serum-free Medium (With phenol red) | EXP0118701 | 1L | Liquid | |||
| HIPP-T006 |
HIPP-T006 Lymphocyte Serum-free Medium (GMP Grade) |
EXP0105301 | 1L | Liquid | ||
|
HIPP-T006 Lymphocyte Serum-free Medium (With phenol red) |
EXP0118801 | 1L | Liquid |
Case Studies
Superior T Cell Expansion With Enhanced Functional Phenotypes Under Serum-free Conditions
Under serum-free conditions, HIPP-X100 demonstrated 1.66-fold and 1.23-fold higher T-cell expansion than Brand T and Brand L, respectively (Day 11), while maintaining >97% viability. Flow cytometry showed a higher CAR+ cell population (72.02%) compared to other brands.
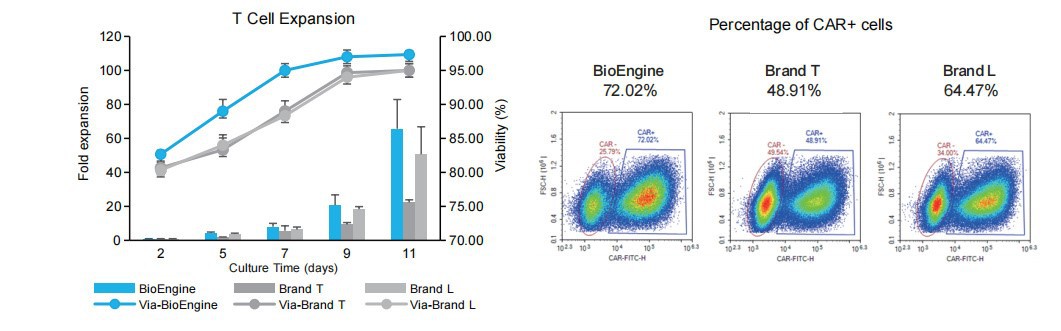
Flow cytometry demonstrated comparable expression of exhaustion markers (PD-1: 20%; TIM-3: 66%; LAG-3: 34%) to other brands, while exhibiting enhanced memory phenotypes (CD45RA+CD62L+: 68.4%; CD45RA+CCR7+: 69.2 %).

Robust NK Cell Expansion Under Serum-free Conditions
In NK cell co-culture with feeder cells, HIPP-T009 outperformed both Brand L serum-free medium (which failed to support NK cell proliferation) and Brand L medium supplemented with 5% FBS. Additionally, HIPP-T009 showed comparable CD3-CD56+ cell percent ages (68.9%) to that in Brand L + 5% FBS culture.

Documents
HIPP Series Serum-free Media
DownloadFAQ
Q1:What is the best media for T cells?
Q2:What are the differences between CAR-T and uCAR-T?
Q3:What is CAR-NK?
Q4:Why is DMEM medium red?
Q5:What are BHK-21 cells?
Q6:Why are serum-free cell culture media needed?
Q7:How is the stability and consistency of cells in serum-free cell culture media?
Previous
No InformationNext
No InformationHot Tags: hipp series serum-free media for immune cell culture,hipp series serum-free media for immune cell culture manufacturers, suppliers, factory
![]() Antibodies
Antibodies
In fed-batch process, CHO cells cultured in Eden CD CHO media demonstrate higher viable cell density (VCD) and protein production than competitors. On average, the antibody titers of various CHO cell lines cultured in Eden CD CHO media ranged from 2 to 11 g/L.

In the pulse perfusion process, CHO cells cultured in Eden CD CHO media demonstrated superior performance than competitive global brands. When VVD=1.0, volumetric productivity (Vp) can reach up to 2.2 g/L/day, and the cumulative product expression in 14 days can reach 19 g/L, 55% higher than global brand B. When VVD=2.0, Vp can reach up to 3.3g/L/day, and the cumulative product expression in 14 days can reach 25 g/L.

Brochures
| CHO Cell Medium |
FAQ
Q1: What is the packaging for BioEngine's powder media?
Q2: Can we seal the bag if there is leftover powdered medium?
Q3: What is the shelf life of BioEngine's Eden series CHO cell culture media, and how do you verify the expiration date?
Q4: Does BioEngine offer any regulators or additives for modulating antibody glycosylation?
Q5: Does BioEngine's CHO cell culture media contain hydrolysates, insulin, cytokines, or other components?
Q6: What are CHO media?
Q7: What are the differences between DMEM and RPMI?
1. Nutrient composition: DMEM (Dulbecco's Modified Eagle's Medium) and RPMI (Roswell Park Memorial Institute) have different nutrient compositions. DMEM contains higher levels of glucose, amino acids, vitamins, and sodium pyruvate, while RPMI has a lower glucose concentration and a different amino acid and vitamin composition.
2. pH: DMEM has a higher pH (7.4-7.6) compared to RPMI (7.2-7.4).
3. Usage: DMEM is a more general-purpose medium and can be used for a wide range of cell types, including adherent and non-adherent cells, while RPMI is typically used for the culture of immune cells such as lymphocytes and hybridomas.
4. Serum requirement: RPMI is often used with lower serum concentrations (e.g. 5-10%) than DMEM, which may require higher serum concentrations (e.g. 10-20%).
Overall, the choice between DMEM and RPMI depends on the specific cell type being cultured and the experimental conditions.
Q8: What is a fed-batch culture?
The goal of a fed-batch culture is to maximize cell growth and productivity while maintaining a stable culture environment. By controlling the rate and timing of nutrient addition, the culture can be kept in a state of controlled growth, avoiding the depletion of nutrients and accumulation of waste products that can limit growth and product formation in batch cultures. Additionally, the use of a fed-batch culture can allow for the accumulation of high cell densities and the optimization of production conditions, leading to higher yields and greater efficiency in bioprocesses.
Q9: What are the advantages of suspension cell cultures?
1. Scalability: Suspension cell cultures can be easily scaled up to produce large quantities of cells. This makes them particularly useful for biomanufacturing and the production of recombinant proteins.
2. Flexibility: Suspension cultures can be adapted to a wide range of culture conditions, such as pH, temperature, and nutrient availability. This allows for the optimization of cell growth and productivity.
3. Homogeneity: Suspension cultures provide a more homogeneous population of cells than adherent cultures, where cells may exhibit varying degrees of differentiation and proliferation.
4. Reduced risk of contamination: Suspension cultures are less prone to contamination by bacteria or fungi than adherent cultures, as there are no surfaces for microorganisms to adhere to.
5. Ease of harvesting: Cells in suspension culture can be easily harvested using centrifugation, filtration, or other methods. This simplifies downstream processing and reduces the risk of damage to the cells.
Overall, suspension cell culture offers several advantages over other types of cell culture, particularly in the context of large-scale biomanufacturing and the production of recombinant proteins.
Q10: What is gene therapy?
●Replacing a disease-causing gene with a healthy copy of the gene.
●Inactivating or deleting a disease-causing gene.
●Introducing a new or modified gene to help fight disease.
Gene therapy is a promising and innovative field of medicine that has potential applications for many diseases, such as cancer, genetic disorders, infectious diseases, and autoimmune diseases 23. However, gene therapy also faces many challenges and risks, such as safety, efficacy, ethical issues, and regulatory hurdles.
Gene therapy usually be delivered to the cells by virus vector, such as AAV, Adv and RV. BioEngine provides vigor series insect cell media for large scale AAV production."
Q11: Does BioEngine provides serum products, like FBS?
Previous
No InformationNext
T Cell MediumSend Inquiry
Provide high quality culture media for research institutions, biopharmaceutical companies and clinical trial institutions, etc.
I agree to receive other communications from BioEngine which may contain technical or marketing information from this organization. You can unsubscribe from these communications at any time.
By submitting this form, you are agreeing to the terms and conditions of our Privacy Policy
You Might Also Like