Chikungunya Vaccines: Only Two Available Worldwide, A Quick Overview Of Key Information
Jan 23, 2026
Leave a message
Outbreaks of chikungunya fever have been reported in multiple regions recently. As a viral infectious disease primarily transmitted by Aedes mosquito bites, it can cause fever, skin rashes, joint and muscle pain, which has attracted widespread public attention. Local authorities have also issued prevention and control measures accordingly. Today, we will learn about chikungunya fever from the perspective of vaccines.
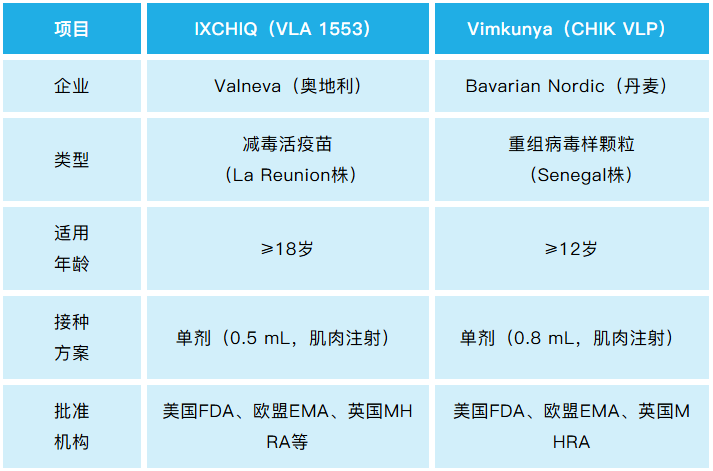
Chikungunya fever is caused by the chikungunya virus. To date, two chikungunya vaccines have been approved for marketing globally, while no such products have been launched in China yet. The two approved vaccines are IXCHIQ (a live-attenuated vaccine) developed by Valneva and Vimkunya (a virus-like particle, VLP vaccine) developed by Bavarian Nordic.
IXCHIQ Vaccine: Approved for marketing in 2023, it is the first chikungunya vaccine and a live-attenuated vaccine produced using Vero cells.
Vimkunya Vaccine: Approved for marketing in February 2025, it is the first chikungunya vaccine indicated for individuals aged 12 years and above, and a VLP vaccine produced using HEK293 cells.
As shown in the figure below, the current chikungunya virus vaccine pipeline covers a variety of technical routes, including live-attenuated/inactivated virus vaccines, protein vaccines, viral vector vaccines, mRNA vaccines and so on.
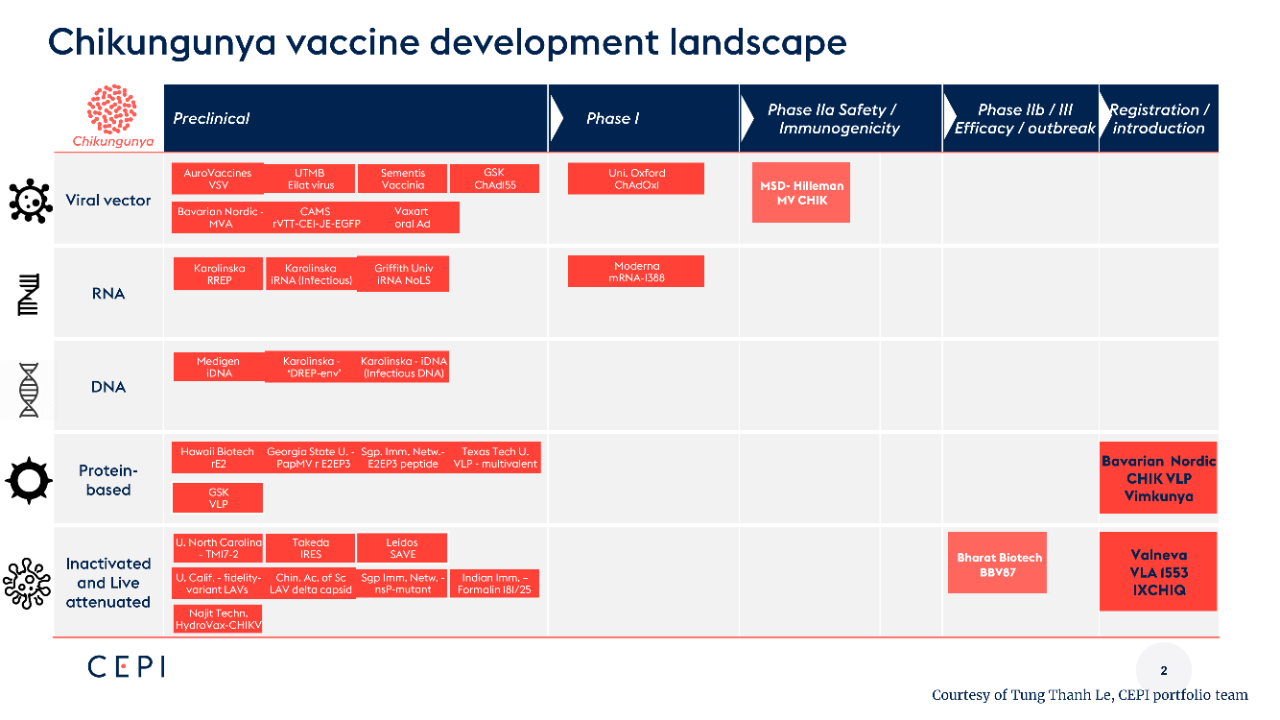
Presentation on Chikungunya Vaccine Development Landscape
- Viral Vectors: Adenovirus, VSV, Poxvirus, etc.
- RNA Vaccines: mRNA, Infectious RNA
- DNA Vaccines: Infectious DNA
- Protein Vaccines: VLP, Recombinant Protein, Polypeptide
- Inactivated/Live-Attenuated Vaccines: Various Subtype Strains, Mutant Strains
Live-Attenuated/Inactivated Virus Vaccines
Live-attenuated/inactivated virus vaccines represent traditional and most common vaccine types. Different viruses require the use of distinct cell platforms. IXCHIQ, the chikungunya vaccine mentioned in this article, is produced using Vero cells.
Derived from the epithelial cells of African green monkey kidneys, Vero cells are commonly used in biologics manufacturing. They exhibit excellent susceptibility and proliferation capacity for a wide range of viruses. Viruses cultured in Vero cells yield high titers, enabling large-scale vaccine production. As a result, such vaccines are relatively cost-effective and have been widely applied.
VLP Vaccines
Virus-like particle (VLP) vaccines are typically composed of pathogen structural proteins, most commonly viral capsid proteins. For instance, VIMKUNYA, as mentioned in this article, contains the capsid protein (C) and envelope proteins E1 and E2 of the chikungunya virus. These proteins self-assemble into virus-mimicking particle structures but do not contain viral genetic material, rendering them non-infectious and thus more suitable for immunocompromised populations. Meanwhile, their structural similarity to native viruses allows them to stimulate the immune system more effectively. However, their production costs are relatively higher compared to the aforementioned live-attenuated/inactivated virus vaccines.
VLP production platforms include Escherichia coli, yeast, insect cell platforms, mammalian cell platforms (e.g., 293, CHO cells), and plant platforms. Previously, we used HPV vaccines as an example to introduce recombinant protein vaccine production platforms and elaborate on the characteristics of different platforms. For details, please refer to our in-depth article: In-depth Insights | The Multi-Billion-Dollar Vaccine Market: Application and Comparison of Different Expression Platforms in HPV Vaccine Production.
Viral Vector Vaccines
Viral vectors inherently possess viral characteristics, allowing them to efficiently infect a variety of cell types. Subsequently, the antigen gene carried by the vector is expressed within the host cells, triggering an immune response. The more well-known adenovirus-vectored vaccines are usually produced using the HEK293 cell platform.
Cell culture constitutes a critical step in the production of live-attenuated/inactivated virus vaccines, recombinant protein vaccines, and adenovirus-vectored vaccines alike. With nearly 40 years of dedicated research in cell-based large-scale vaccine production, the R&D team of BioEngine has established a strong research background and extensive project experience. We have successfully launched over 150 serum-free medium products, which basically cover all cell culture media required for R&D and production across the entire vaccine field, including the Vero cells, insect cells, HEK293 cells, and CHO cells mentioned in this article.

