Product Introduction: Eden Series CHO Cell CD Medium
Jan 29, 2026
Leave a message
Eden Series Products
Product Features
Serum-free, protein-free, animal-origin-free, with a chemically defined composition
Suitable for CHO-K1, CHOZN, Horizon, CHO-S, CHO-DG44 and other cell lines
Enables high-efficiency antibody/protein expression in various culture processes including fed-batch, high-density fed-batch and perfusion
Product Advantages
Higher Protein Titer
The Eden series medium delivers an average 30% increase in antibody/protein titer, with a maximum titer of up to 14g/L in fed-batch culture, significantly reducing customers' production costs.
Flexible Glycosylation Modulation
Optimized medium components related to glycosylation modification enable fine-tuning of glycosylation profiles. A full range of glycosylation modulators are also available to meet diverse application requirements.
Compliance with Global Regulatory Standards
Certified to ISO13485:2016 and MDSAP; fully compliant with regulatory requirements of Europe, the US and other major regions. A lean and digital management system ensures complete data traceability, providing robust support for IND and BLA filings.
Stable and Reliable Quality
Raw materials are sourced from a rigorously selected supply chain of 2 local + 1 imported qualified suppliers, ensuring reliable quality and stable supply. Advanced CMPM manufacturing processes guarantee excellent batch-to-batch consistency (CPK>1.33, RSD<10%).
Professional Technical Support
Our technical support team boasts profound professional expertise and extensive project experience, providing customers with comprehensive technical support and customized solutions.
Product Performance
Culturing CHO cells in conventional fed-batch processes with the Eden series CHO Cell CD Medium results in significantly higher cell growth rate, maximum viable cell density and viable cell rate at harvest compared to peer brands.
Protein expression titer is doubled in comparison with imported peer Brand A. Meanwhile, critical quality attributes including product charge variant distribution and fragmentation all meet customer specifications, achieving substantial reduction in production costs.
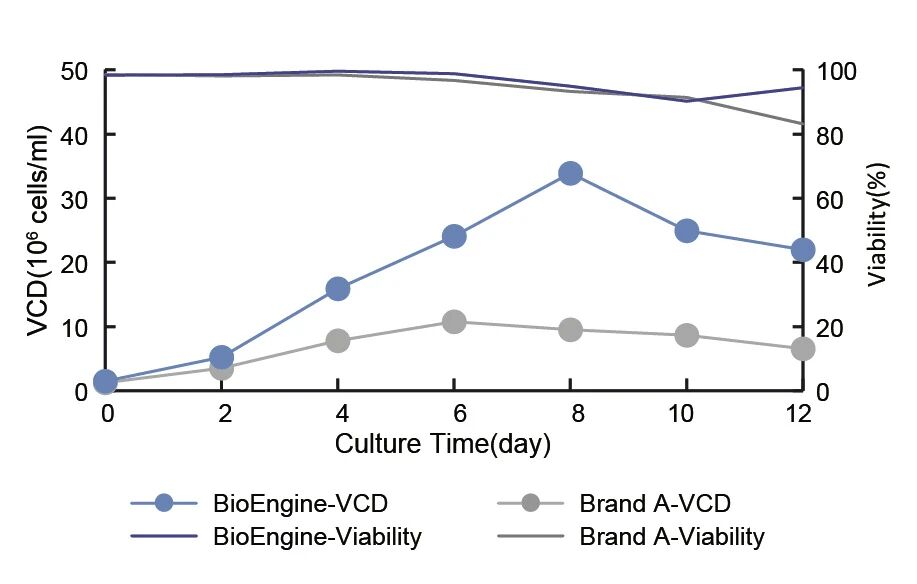
The cell growth rate, maximum viable cell density, and viable cell rate at harvest are all significantly higher than those of imported peer Brand A.

Critical quality attributes such as product charge variant distribution and fragmentation are comparable to those of imported peer Brand A.
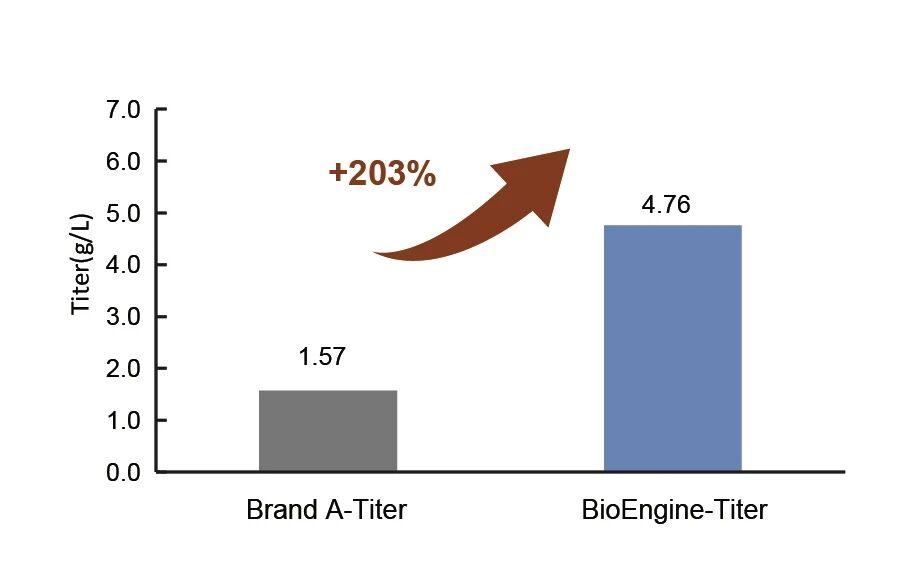
Protein expression titer is tripled compared to that of imported peer Brand A.

