In-depth Insights | Strategies For Regulating Antibody Charge Heterogeneity: From Molecular Mechanisms To Process Optimization
Jan 23, 2026
Leave a message
In late April, we hosted a live webinar titled "Panda Class – Special Topic on Antibody Quality Control and Testing". After the session, we received feedback from some participants, who expressed a desire for more in-depth discussions on content in this field.
To address this demand, we are launching a thematic series on antibody drug quality control. In this first installment, we will focus on the regulation of charge heterogeneity. During antibody drug production, cell culture processes and medium components are crucial to product quality, as they affect the function, structure, and stability of the final product. Charge heterogeneity is usually associated with post-translational modifications and degradation of proteins, including deamidation, oxidation, glycation, glycosylation, and C-terminal lysine cleavage, among others.
Acidic Charge Variants
The formation of acidic variants is linked to deamidation, glycation, and other reactions. Oxidative stress in cell culture (e.g., hydrogen peroxide accumulation and elevated reactive oxygen species (ROS)) can significantly increase monoclonal antibody glycation, which in turn leads to a rise in acidic charge variants (ACVs).
Therefore, oxidative stress can be reduced to decrease ACV levels by optimizing culture processes (such as lowering culture temperature) and medium components (adding antioxidants like iron ions and rosmarinic acid). Studies also indicate that long-term storage of culture medium can cause nutrient degradation and peroxide accumulation, thereby increasing the risk of ACV formation. For this reason, strict control over the storage duration of fresh medium is required.
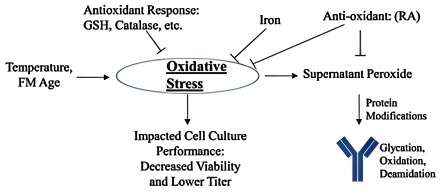
Schematic Diagram 1 of Molecular Mechanisms Affecting the Generation of Acidic Variants
In the formulation design of serum-free media, BioEngine not only focuses on the functions of individual components but also comprehensively considers the stability of each component, thereby controlling issues related to charge heterogeneity at the source.
Acidic Charge Variants
Basic variants are typically associated with the retention of C-terminal lysine, proline amidation, and the cyclization of N-terminal glutamic acid (Glu). For example, the cyclization of N-terminal glutamic acid (Glu) to pyroglutamic acid (pyroGlu) is a common post-translational modification in antibodies, which actually generates a basic variant. Attention should be paid to the culture and storage conditions (such as temperature and pH) to slow down the cyclization reaction².
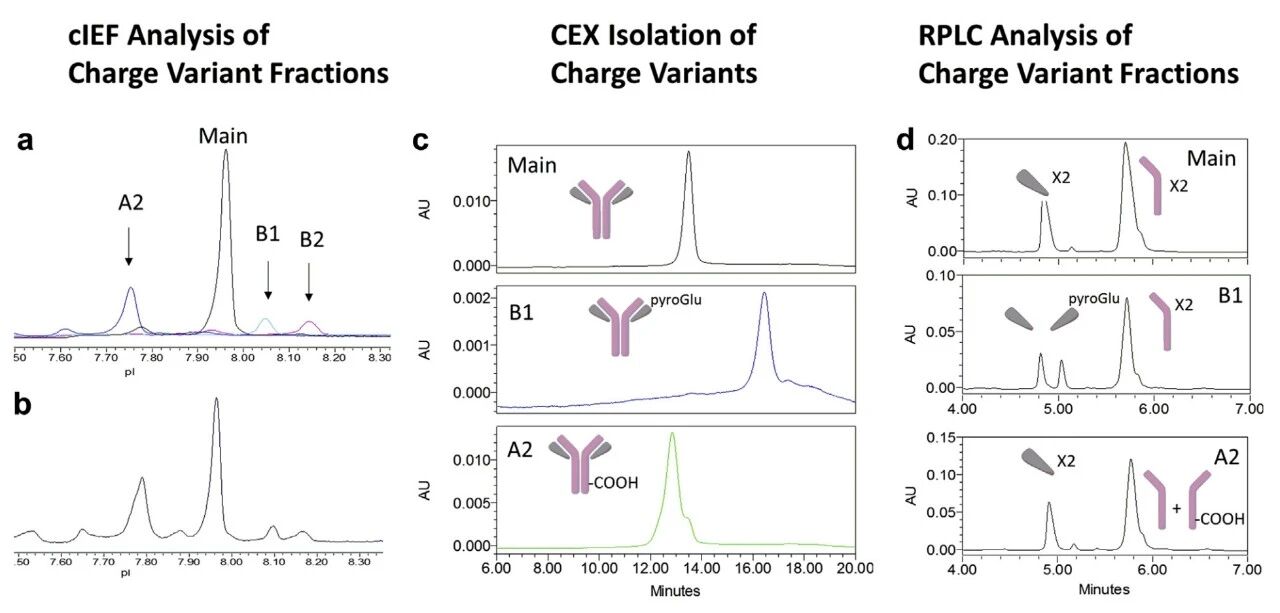
Characterization of Monoclonal Antibody Charge Variants²
Copper ions, which are also a concern for many, have long been a research hotspot regarding their role in antibody production. An appropriate concentration of copper ions can enhance cell viability and antibody yield, while an excessively high concentration will promote C-terminal proline amidation, thereby increasing the level of basic variants. Therefore, optimizing medium formulations and process parameters can effectively reduce the negative impact of copper ions on charge heterogeneity³.
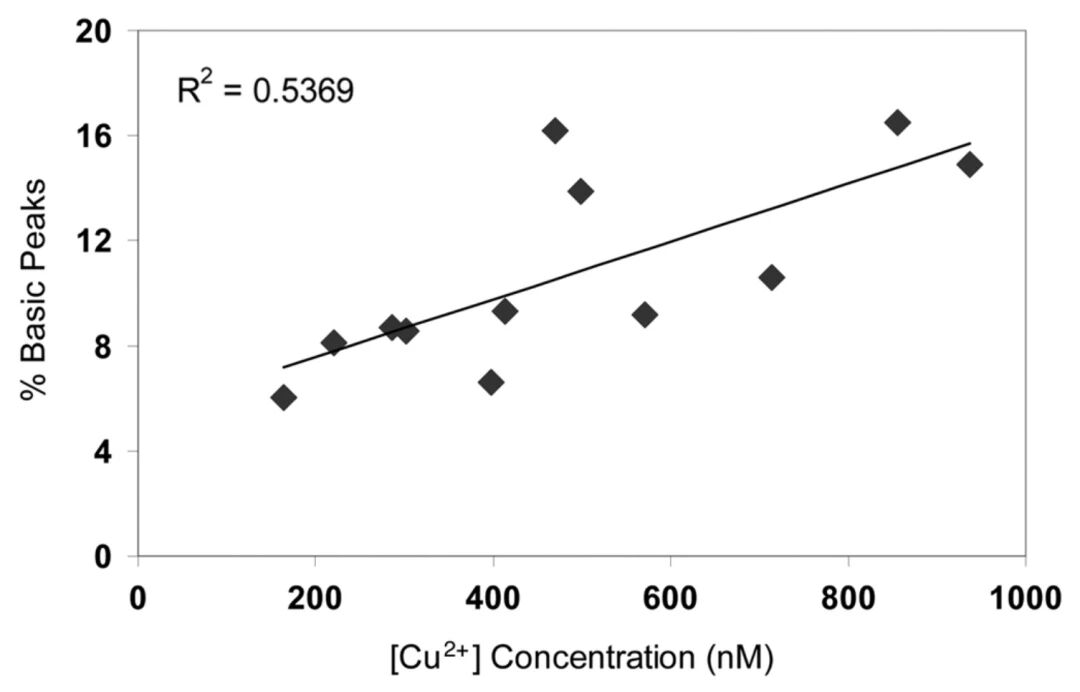
Correlation Between Copper Ion Concentration and Basic Peak Content³
As a professional supplier of serum-free cell culture media, Bainaji has been successfully applied in over 120 antibody/protein drug pipelines, and accumulated rich experience in addressing critical quality attribute (CQA) issues such as charge heterogeneity and glycosylation profiles. Bainaji offers a comprehensive solution for CHO cell culture media, including core products such as transient protein expression kits, serum-free media for cell line development, fed-batch medium kits for large-scale production, and protein quality regulators. In addition to products, Bainaji also provides customized medium formulation development and large-scale cell culture technical services to meet your diverse personalized requirements.
For more literature interpretations and practical experience related to cell culture, please follow the Bainaji official WeChat account.
[Reference]
Chung S, Tian J, Tan Z, et al. Modulating cell culture oxidative stress reduces protein glycation and acidic charge variant formation. mAbs. 2019;11(1):205-216. doi:10.1080/19420862.2018.1537533
Liu Z, Valente J, Lin S, Chennamsetty N, Qiu D, Bolgar M. Cyclization of N-Terminal Glutamic Acid to pyro-Glutamic Acid Impacts Monoclonal Antibody Charge Heterogeneity Despite Its Appearance as a Neutral Transformation. J Pharm Sci. 2019;108(10):3194-3200. doi:10.1016/j.xphs.2019.05.023
Kaschak T, Boyd D, Lu F, et al. Characterization of the basic charge variants of a human IgG1: Effect of copper concentration in cell culture media. mAbs. 2011;3(6):577-583. doi:10.4161/mabs.3.6.17959

